Superfund Research Program
Tracking Mercury Conversion and Distribution in Aquatic Environments
Release Date: 04/03/2024
![]() subscribe/listen via iTunes, download(7.9MB), Transcript(65KB)
subscribe/listen via iTunes, download(7.9MB), Transcript(65KB)
NIEHS Superfund Research Program (SRP)-funded researchers, led by Heileen Hsu-Kim, Ph.D., of the Duke University SRP Center, provided insight into how and at what timescale mercury changes within a wetland ecosystem. They found mercury from different sources is converted into other mercury forms that eventually have similar properties. This finding can inform environmental management or pollution control strategies.
Mercury, a metal that is toxic to living organisms, can be found in the environment in several forms with distinct properties and toxicity levels. Methylmercury, one of the metal’s most toxic forms, can build up in the tissues of fish, posing a threat to humans through consumption.
Mercury often enters aquatic ecosystems in multiple ways and in many forms, such as particulate mercury from the erosion of contaminated soils and dissolved mercury deposited into surface waters from the atmosphere. Upon entering an aquatic ecosystem, these different species of mercury are exposed to the same environmental processes and converted into other forms of mercury with almost identical properties and reactivity to one another. This process is known as homogenization.
Creating Miniature Ecosystems

The team aimed to understand how and when mercury homogenization occurs in the environment. They first constructed three miniature freshwater wetlands in boxes outdoors. Each box consisted of sediment and water, as well as organisms typical of such environments, including several types of plants and a common freshwater fish known as mosquitofish.
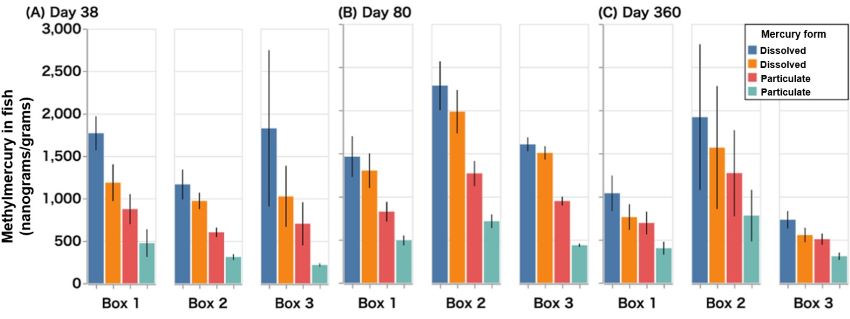
The researchers gave the wetlands three months to establish, then they added four different forms of mercury: two types of particulates, simulating mercury from suspended soils; and two dissolved forms, mimicking water and atmospheric deposition sources of mercury. The team collected surface water and sediment samples from each wetland at three, 28, 81, and 360 days after the initial addition of mercury. They also collected adult mosquitofish at 38, 80, and 360 days, to analyze how different mercury species are taken up by aquatic organisms.
Revealing Trends
Analysis of water samples showed that, for all three wetlands, the four forms of mercury were relatively similar in concentration by the third day. But, by day 38, mercury concentrations were greater for the two initially dissolved mercury species than the initially particulate mercury, and this trend continued for the rest of the experiment. Overall, concentrations of dissolved and particulate mercury in water remained different throughout the experiment duration.
In contrast, in sediments, all four forms reached a relatively similar concentration and achieved homogeneity within two months. Interestingly, before the two-month mark, sediment mercury concentrations differed greatly between boxes at several time points. The authors explained that, while the three experiments were set up similarly, these results indicate that different ecosystems undergo unique biological processes that control their chemical components.
The team also analyzed how much of the mercury from each species was converted to methylmercury and how much methylmercury was taken up by mosquitofish. They observed that, at all sampling points, methylmercury concentrations in sediments and fish were five times greater for the dissolved mercury forms compared to the particulate mercury.
Environmental Implications
The scientists also deployed passive sampling devices, previously developed by the team, for seven days at three sampling points: 17, 38, and 360 days following initial mercury addition.
They observed that in both surface water and sediment, mercury concentrations quantified by the passive samplers were higher for initially dissolved mercury forms compared to particulate mercury. Dissolved and particulate mercury concentrations remained differentiated throughout the study and started to converge toward similar values at the one-year mark. According to the authors, these results suggest that, for sediments, mercury concentrations collected by their sampling device can remain differentiated longer than concentrations from traditional sampling methods, providing a better picture of sources of mercury contamination.
This study provides insight into the timescale needed for different mercury sources to reach homogeneity in composition and reactivity in aquatic ecosystems, the team said. Water quality management is challenging because strategies often focus on controlling mercury releases from specific contamination sources, such as wastewater and emissions from waste incinerators. The researchers emphasize the importance of evaluating both mercury sources and the processes mercury may undergo when devising strategies to manage contamination.
For More Information Contact:
Heileen Hsu-Kim
Duke University
Civil and Environmental Engineering
Box 90287
Durham, North Carolina 27708-0287
Phone: 919-660-5109
Email: hsukim@duke.edu
To learn more about this research, please refer to the following sources:
- Wadle A, Neal-Walthall N, Ndu U, Hsu-Kim H. 2024. Distribution and homogenization of multiple mercury species input to freshwater wetland mesocosms. Environ Sci Technol doi:10.1021/acs.est.3c07169 PMID:38181227
To receive monthly mailings of the Research Briefs, send your email address to srpinfo@niehs.nih.gov.


