Superfund Research Program
Sampling Device May Predict Methylmercury Accumulation in Wetlands
View Research Brief as PDF(405KB)
Release Date: 04/06/2022
![]() subscribe/listen via iTunes, download(5.6MB), Transcript(108KB)
subscribe/listen via iTunes, download(5.6MB), Transcript(108KB)
NIEHS Superfund Research Program (SRP)-funded researchers, led by Heileen Hsu-Kim, Ph.D., of the Duke University SRP Center, showed that a small plastic sampling device can efficiently predict the potential for methylmercury — an environmental contaminant — to form in freshwater wetlands and to accumulate in organisms living there.
The contaminant can build up in animal tissues, with toxic effects to the nervous system. Because methylmercury occurs in greater amounts up the food chain, humans who eat large fish can also be exposed.
In aquatic ecosystems, inorganic mercury changes to methylmercury through microbial activity. Inorganic mercury occurs naturally in the environment but can also be found in some consumer products, as well as in emissions from coal-fired power plants and other industrial activities.
Making Miniature Wetlands
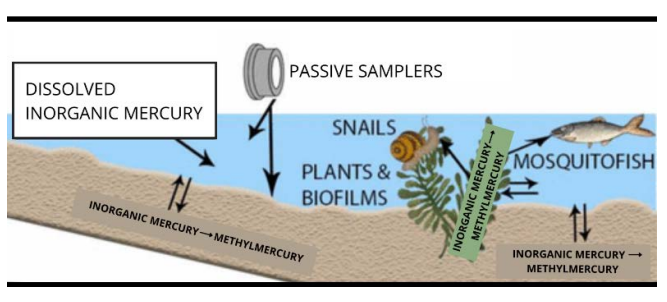
The researchers' sampling device consists of three main parts:
- A filter that sieves the surrounding environment.
- A gel through which matter spreads to different extents based on its size.
- A resin layer that binds that material.
To test the sampler, they first constructed six miniature freshwater wetlands in boxes outdoors. Each box consisted of sediment and water, as well as organisms typical of such environments.
The team gave the wetlands three months to establish, then collected sediment and water samples to measure their baseline mercury concentrations.
Next, to half of the boxes, the researchers added several solutions, each containing a traceable form of inorganic mercury that was either dissolved or particulate — stuck to small pieces of matter. The remaining boxes served as controls.
The team allowed the mercury to work its way through the wetland systems, then deployed the samplers, some face-down in the sediment and others suspended in the water column, for a week. The idea was to see how much inorganic mercury became methylated, where it ended up, and how useful the samplers were at predicting those changes.
After retrieving the devices, the researchers again collected water and sediment samples, as well as mosquitofish, snails, plants, and biofilms — communities of microorganisms that cling to submerged surfaces — for laboratory analysis.
Analyzing Trends
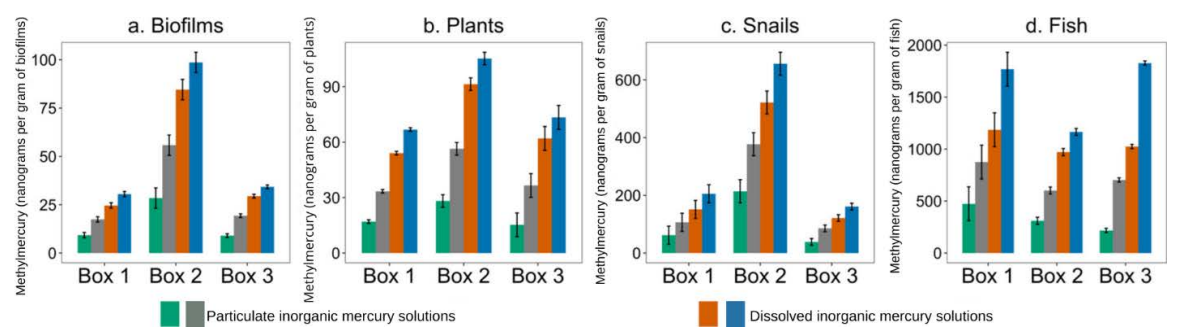
When the researchers analyzed their sediment sampler data, they found that inorganic mercury concentrations were moderately predictive of methylmercury accumulation in that sediment.
They also found that methylmercury measured by the water samplers correlated strongly with methylmercury accumulation in the biological samples. But an even better predictor of methylmercury content in organisms was total mercury — inorganic mercury plus methylmercury — measured by those samplers.
That finding could reflect pathways for methylmercury buildup in the food chain, the authors noted. For example, biofilms can convert inorganic mercury into methylmercury, as well as absorb the toxin directly. Those same biofilms could also be an important food source for snails and fish in wetlands. In addition, aquatic plants could absorb methylmercury produced by biofilms living on them.
According to the authors, the study shows how their samplers can be used both to predict the potential of inorganic mercury to transform into methylmercury in freshwater wetlands and to quantify the threat posed to organisms living there. Further, their findings reveal the importance of sampling water instead of sediment.
The authors added that future efforts to monitor mercury in aquatic ecosystems should consider the activity of microbes involved in methylmercury formation and decomposition.
For More Information Contact:
Heileen Hsu-Kim
Duke University
Civil and Environmental Engineering
Box 90287
Durham, North Carolina 27708-0287
Phone: 919-660-5109
Email: hsukim@duke.edu
To learn more about this research, please refer to the following sources:
- Neal-Walthall N, Ndu U, Rivera NA, Elias DA, Hsu-Kim H. 2022. Utility of diffusive gradient in thin-film passive samplers for predicting mercury methylation potential and bioaccumulation in freshwater wetlands. Environ Sci Technol 56:doi:10.1021/acs.est.1c06796 PMID:35044747
To receive monthly mailings of the Research Briefs, send your email address to srpinfo@niehs.nih.gov.


