Superfund Research Program
Collaborative Cross Mice Can Fill Data Gaps in Risk Assessment
View Research Brief as PDF(470KB)
Release Date: 10/02/2019
![]() subscribe/listen via iTunes, download(9.5MB), Transcript(90KB)
subscribe/listen via iTunes, download(9.5MB), Transcript(90KB)
NIEHS Superfund Research Program (SRP) grantees showed how the Collaborative Cross (CC) mouse model, which uses genetically diverse mice to capture over 90% of known mouse genetic variations, can account for individual differences in susceptibility to environmental chemicals. Led by Ivan Rusyn, M.D., Ph.D., director of the Texas A&M University SRP Center, researchers measured variability in kidney toxicity and metabolism in CC mice after exposing them to tetrachloroethylene (PERC). For kidney toxicity mediated by this chemical, they found that default risk assessment factors used to account for human variability may be marginally adequate to protect 95%, but not more, of the population.

PERC, also known as PCE, is a solvent frequently used in dry cleaning solutions, adhesives, and metal degreasers. It can be released into the air, water, and soil at places where it is produced or used, and it is commonly found in groundwater or soil at hazardous waste sites.
Using PERC as a Case Study
People with different genetic make-up may metabolize chemicals differently, making them more or less likely to experience harmful health effects based on how those metabolites are distributed within the body and how they interact with cells and organs.
PERC can be broken down by the body in several different ways, which can vary between individuals and lead to organ-specific toxicity associated with several different metabolic pathways. Trichloroacetic acid (TCA) is a major metabolite of PERC, which may contribute to liver toxicity. On the other hand, kidney toxicity of PERC is linked to metabolites related to glutathione (GSH) conjugation, a pathway that is generally involved in detoxifying chemicals in the body.
To estimate differences in PERC toxicity, the research team characterized variability in PERC metabolism and kidney toxicity in male mice from 45 CC strains. Overall, mice exposed to PERC had lower kidney weight and more markers of kidney injury, supporting previous findings that the kidney is a target of PERC toxicity. They also linked specific GSH conjugates to kidney injury markers, indicating that GSH conjugation contributes to this toxicity.
Among the 45 CC strains tested, the team found variation in PERC metabolism and toxicity, with greater variations observed in kidney toxicity, including differences in kidney weight and different levels of genes and proteins known to be associated with kidney injury. The findings show that some mouse strains, including the C57BL/6J mouse strain commonly used in research, do not have observable kidney effects following PERC exposure. However, in the CC population-based model, PERC has overall effects on kidney injury.
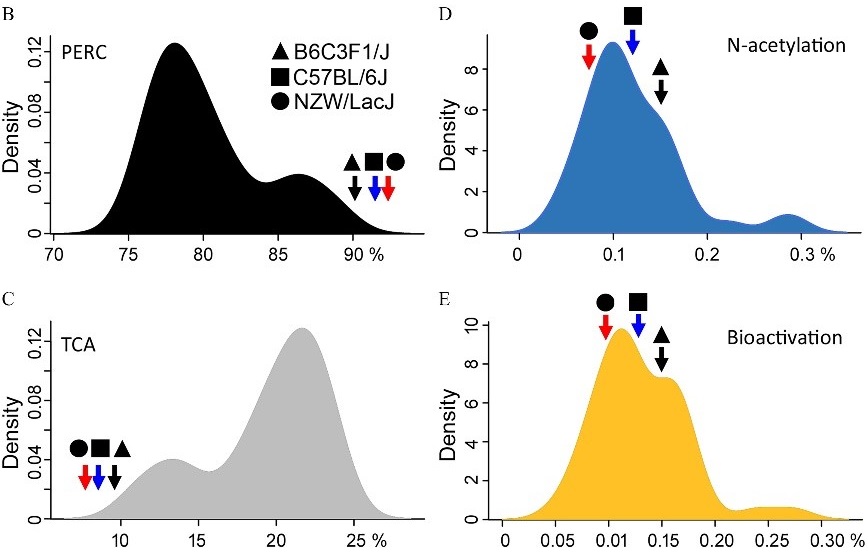
The researchers estimated the amount of PERC and its metabolites that would remain in tissues after exposure. Once deposited in tissues, these chemicals may lead to additional toxicity. Deposition varied within the population, with an average of 80.4% for PERC, 19.4% for TCA, and 0.25% for GSH conjugates. Interestingly, inbred mice used in previous studies generally exhibited GSH conjugation of PERC in the middle of the CC distribution, while their metabolism of PERC is at the lower end of the CC distribution. Because differences in metabolism can affect how quickly toxicants leave the body, lower metabolism of PERC may indicate that those inbred strains may underestimate effects in a more general population.
According to the authors, this research shows that the CC mouse population approach provides a better characterization of population variability in metabolism of PERC than previously demonstrated with a few inbred mouse strains.
Informing Risk Assessment
Regulatory agencies use human health risk assessments to determine safety thresholds for exposures to harmful chemicals, often calculated based on studies in rodents. To protect the general human population, uncertainty factors are applied, which lower the safety threshold to account for data limitations, such as differences between humans and rodents and differences between individuals in a population.
According to the authors, when comparing the results of this study to the default values generally used in human health risk assessments to adjust for population variation, the default values may not protect the most sensitive individuals. Because of population variation in metabolism and toxicity, the authors add that the traditional risk assessment approach of combining metabolism and toxicity variability by simple multiplication may not be the best approach. Separating the variability into two different components, using population-based models like CC strains, may provide a more accurate estimate.
This study illustrates how the CC mouse population can be used to fine-tune protective adjustment factors for population variability in risk assessments. Moving forward, the research team aims to update models that predict the distribution, metabolism, and elimination of environmental chemicals in the body with population-based mouse data to provide more refined metabolite- and tissue-specific predictions of PERC toxicity.
For More Information Contact:
Ivan Rusyn
Texas A&M University
273 VRBA
College Station, Texas 77843-4458
Phone: 979-458-9866
Email: irusyn@cvm.tamu.edu
To learn more about this research, please refer to the following sources:
- Luo Y, Cichocki JA, Hsieh N, Lewis L, Wright FA, Threadgill DW, Chiu WA, Rusyn I. 2019. Using Collaborative Cross mouse population to fill data gaps in risk assessment: A case study of population-based analysis of toxicokinetics and kidney toxicodynamics of tetrachloroethylene. Environ Health Perspect 127(6):67011. doi:10.1289/EHP5105 PMID:31246107 PMCID:PMC6792382
To receive monthly mailings of the Research Briefs, send your email address to srpinfo@niehs.nih.gov.


