Superfund Research Program
New Model to Examine PFAS Sheds Light on Lipid Disruption Mechanisms
View Research Brief as PDF(489KB)
Release Date: 01/13/2021
![]() subscribe/listen via iTunes, download(6.9MB), Transcript(138KB)
subscribe/listen via iTunes, download(6.9MB), Transcript(138KB)
Researchers from the Boston University (BU) Superfund Research Program (SRP) Center developed a novel study design that generated new insight on the effects of perfluorooctanoic acid (PFOA) on cholesterol regulation in the liver. Led by Jennifer Schlezinger, Ph.D., the team also investigated the effects of PFOA on the human peroxisome proliferator activated receptor α (hPPARα), a transcription factor that regulates lipid homeostasis.
PFOA is a fluorinated organic compound that is part of the larger group of compounds known as perfluoroalkyl substances (PFAS). Adverse health outcomes related to birth, immunologic effects, and metabolic disruption have been associated with exposure to these substances.
While both mouse PPARα (mPPARα) and human PPARα (hPPARα) have been shown to contribute to regulation of cholesterol homeostasis, there are well known species differences in the function of mPPARα and hPPARα. Past studies of the effects of PFOA on lipids and cholesterol levels in animal models have produced contradictory results. Depending on variables like sex and diet, mice exposed to PFOA can show either decreased or increased total cholesterol levels in contrast to the increase shown in human epidemiology.
With this in mind, the researchers examined an untested combination of variables.
A Unique Combination
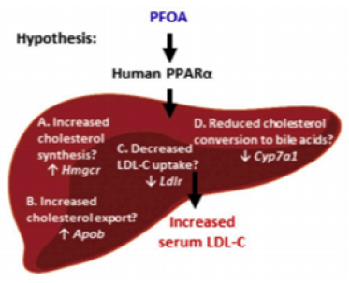
The BU researchers tested the hypothesis that PFOA exposure dysregulates genes controlling cholesterol homeostasis through four potential pathways in liver:
- Increased cholesterol synthesis.
- Increased cholesterol export into the blood.
- Decreased hepatic uptake of Low-density lipoprotein (LDL-C) from blood.
- Decreased cholesterol turnover to bile acids.
Female and male hPPARα and PPARα null mice were fed a high fat/high cholesterol diet based on the “what we eat in America” analysis diet and were exposed to PFOA in drinking water for six weeks.
The team chose this diet because standard rodent chow is generally low in fat with negligible cholesterol. After six weeks the researchers measured serum cholesterol and analyzed the mouse livers, an essential site of regulation of multiple aspects of cholesterol homeostasis.
Multiple linear regression modeling was used to investigate the contribution of sex and genotype on the outcomes. PFOA increased liver mass in both sexes and histological analysis showed lipid accumulation. Their analyses of serum lipoprotein cholesterol suggest that PFOA increased serum cholesterol, particularly in male mice.
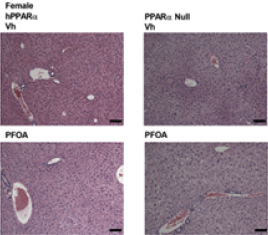
The authors concluded that PFOA modulates at least the PPARα, PPARγ and constitutive androstane receptor (CAR) pathways in liver of hPPARα mice, as well as multiple genes involved in cholesterol metabolism and homeostasis. PPARγ is another member of the PPAR subfamily of nuclear receptors and CAR is a transcriptional regulator of certain metabolic functions. They also found that not all effects were PPARα-dependent and that the hepatic response to PFOA exposure is sexually dimorphic.
Essential New Information
The research provides essential new information to understand the mechanism(s) by which PFAS can affect the amount of lipids in blood. The crucial role of hPPARα in basal cholesterol homeostasis, as well as fatty acid homeostasis, and known species differences in ligand binding gene components support the BU researcher’s conclusion that this model is an important new tool in exploring multiple, interacting mechanisms of PFOA action on cholesterol homeostasis.
Additionally, a previous study investigated the effects of PFAS on lipid homeostasis in female mice. The results corroborate the observation of sex-dependent effects of PFOA on liver physiology.
The question remains whether dyslipidemia, an abnormal amount of lipids in the blood, is a critical effect of PFAS. But the results from this study, and future studies that use this model, can provide information to understand the mechanism(s) by which PFAS could induce dyslipidemia. Importantly, PFOA-induced effects appear to be sex-dependent, and further research is needed to delineate the biologically significant effects of PFAS on multiple aspects of cholesterol homeostasis.
For More Information Contact:
Jennifer J Schlezinger
Boston University
Department of Environmental Health
Sherr Lab, R4
Boston, Massachusetts 02118
Phone: 617-358-1708
Email: jschlezi@bu.edu
Thomas F Webster
Boston University
Department of Environmental Health
Talbot, 4W
Boston, Massachusetts 02118
Phone: 617-638-4620
Email: twebster@bu.edu
To learn more about this research, please refer to the following sources:
- Schlezinger JJ, Puckett H, Nielsen OG, Heiger-Bernays WJ, Webster TF. 2020. Perfluorooctanoic acid activates multiple nuclear receptor pathways and skews expression of genes regulating cholesterol homeostasis in liver of humanized PPAR alpha mice fed an American diet. Toxicol Appl Pharmacol 405:115204. doi:10.1016/j.taap.2020.115204 PMID:32822737 PMCID:PMC7503133
To receive monthly mailings of the Research Briefs, send your email address to srpinfo@niehs.nih.gov.


